An investigational therapy from Alnylam Pharmaceuticals did not significantly lower blood pressure in a key Phase 2 trial, raising questions about the benefit of the twice-a-year treatment as it enters late-stage testing.
Among high-risk patients already taking at least two common hypertension treatments, a 300-mg dose of Alnylam’s zilebesiran led to a reduction in blood pressure of 5 millimeters of mercury (mmHG) lower than placebo at three months. By six months, the difference was 3.9 mmHG. However, these results were not considered statistically significant, according to data released Saturday at a meeting of the European Society of Cardiology.
Researchers also tested a higher dose 600 mg of zilebesiran, but didn’t observe additional benefits.
To read the rest of this story subscribe

 STAT News
STAT News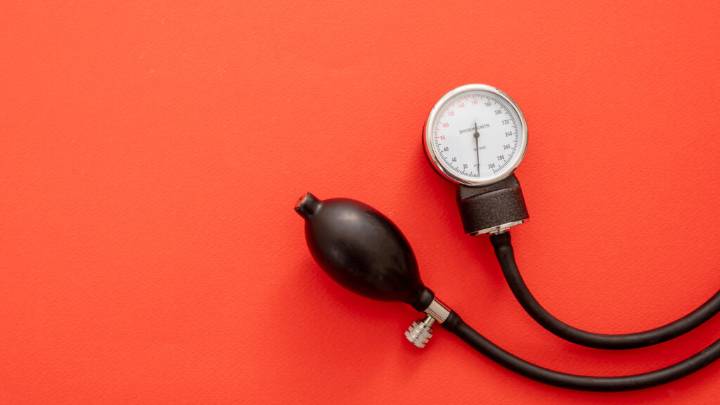

 Reuters US Domestic
Reuters US Domestic Raw Story
Raw Story AlterNet
AlterNet NBC News
NBC News The US Sun Health
The US Sun Health America News
America News WKOW 27
WKOW 27 The Daily Sentinel
The Daily Sentinel New York Magazine
New York Magazine NBC Sports Soccer
NBC Sports Soccer